János TÓTH1, Krisztián KOVÁCS2, Béla VIZVÁRI3, Miklós RIEDEL2
1 Department of Mathematical Analysis, Budapest University
of Technology and Economics
H-1111 Budapest, Egry J. u. 1., HUNGARY
2 Department of Physical Chemistry, Loránd
Eötvös University
H-1117 Budapest, Pázmány P. sétány 1/ A.,
HUNGARY
3Department of Operations Research, Loránd
Eötvös University
H-1117 Budapest, Pázmány P. sétány l/C.,
HUNGARY
The aim of the present work
is to decompose a complicated chemical reaction into elementary steps
with full knowledge of the species figuring in the process. The elaborated
computational method also permits to arrange reaction steps into right
order therefore a complete reaction mechanism can be established. As all
the possible decompositions can be generated, there is chance of finding
mechanisms inaccessible to the chemical intuition.
The many times investigated but still elusive permanganate/oxalic acid
reaction serves as an example to demonstrate the performance of the method.
The autocatalytic reaction
atomic matrix of the species
|
|
|
|
|
|
|
| H2C2O4 |
|
|
|
|
|
| HC2O4- |
|
|
|
|
|
| H+ |
|
|
|
|
|
| C2O42- |
|
|
|
|
|
| CO2- |
|
|
|
|
|
| CO2 |
|
|
|
|
|
| H2O |
|
|
|
|
|
| MnO4- |
|
|
|
|
|
| MnO2 |
|
|
|
|
|
| Mn3+ |
|
|
|
|
|
| MnC2O4 |
|
|
|
|
|
| Mn2+ |
|
|
|
|
|
| [MnC2O4, MnO4-, H+] |
|
|
|
|
|
| [MnC2O42+, MnO3-]+ |
|
|
|
|
|
| [MnC2O42+, MnO3-, H+]2+ |
|
|
|
|
|
| [H+, MnO2, H2C2O4]+ |
|
|
|
|
|
| [MnO2, H2C2O4] |
|
|
|
|
|
| [Mn(C2O4)]+ |
|
|
|
|
|
| [Mn(C2O4)2]- |
|
|
|
|
|
Computation of reaction steps
Requirements
1. The steps fulfil the law of atomic balance and also the law of charge
balance.
2. Elementary reaction step is either uni- or bimolecular
3. Reversible steps are considered as pairs of reaction and antireaction
(i.e. they are two steps)
We are going to construct all the possible uni- and bimolecular steps
fulfilling the first requirement.
Levels of species and reaction steps
There is another necessary condition to be fulfilled. At the very beginning
of the reaction only reaction steps among the initial species can take
place. Then, reaction steps using the products of these reactions can also
enter the scene, and so on. This hierarchy can be formalised as follows.
|
|
|||
|
|
|
colour | |
| species |
|
|
code |
| MnO4- |
|
|
|
| H2O |
|
|
|
| H2C2O4 |
|
|
|
| H+ |
|
|
|
| MnC2O4 |
|
|
|
| MnO2 |
|
|
|
| Mn2+ |
|
|
|
| H2C2O4- |
|
|
|
| CO2 |
|
|
|
| CO2- |
|
|
|
| [MnO2, H2C2O4] |
|
|
|
| [Mn(C2O4)2]- |
|
|
|
| [Mn(C2O4)]+ |
|
|
|
| C2O42- |
|
|
|
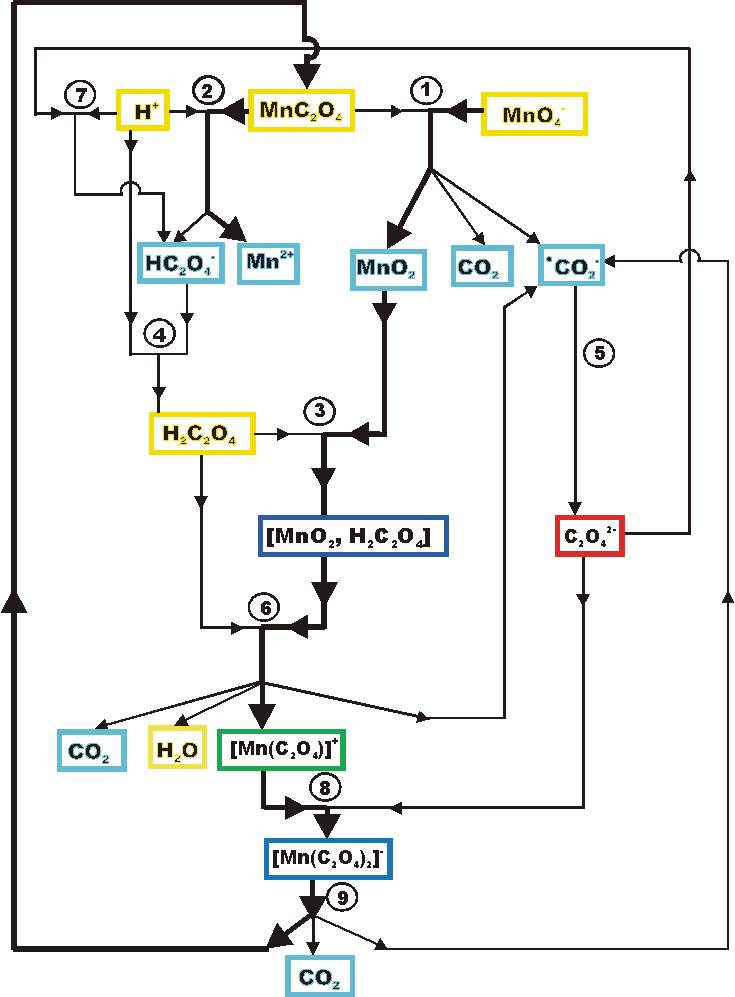
It is obvious (1) that one has to adjoin MnC2O4 to the set of initial species. The new and final computation result (2) shows that the sequence of the reaction steps is acceptable.
Chemically interpreted computational results 1
By the algorithm we obtained 249 decompositions of the overall reaction.
The following is chosen to demonstrate the course of the levelling process.
Levelling 1
equation
H2C2O4 + MnO2 ®
[MnO2, H2C2O4]
(3)
H2C2O4 + [MnO2, H2C2O4]
®
CO2- + CO2
+ 2H2O + [Mn(C2O4)]+
(6)
H+ + HC2O4- ®H2C2O4
(4)
C2O42- + H+ ®
HC2O4-
(7)
H+ + MnC2O4 ®HC2O4-
+ Mn2+
(2)
C2O42- + [Mn(C2O4)]+®
[Mn(C2O4)2]-
(8)
MnC2O4 + MnO4- ®
CO2- + CO2 + 2 MnO2
(1)
[Mn(C2O4)2]- ®
CO2- + CO2 + MnC2O4
(9)
2 CO2- ® C2O42-
(5)
Repeated computation also with MnC2O4 as initial
species
Levelling 2
index
equation
MnC2O4 + MnO4- ®
CO2- + CO2 + 2 MnO2
0
(1)
H+ + MnC2O4 ®HC2O4-
+ Mn2+
0
(2)
H2C2O4 + MnO2 ®
[MnO2, H2C2O4]
1
(3)
H+ + HC2O4- ®H2C2O4
1
(4)
2 CO2- ® C2O42-
1
(5)
H2C2O4 + [MnO2, H2C2O4]
®
CO2- + CO2
+ 2H2O + [Mn(C2O4)]+
2
(6)
C2O42- + H+ ®
HC2O4-
2
(7)
C2O42- + [Mn(C2O4)]+®
[Mn(C2O4)2]-
3
(8)
[Mn(C2O4)2]- ®
CO2- + CO2 + MnC2O4
4
(9)
Discussion

Perspectives
By this computation method